 These billions of atoms can’t realistically be visualized or represented in a timely fashion, yet scientists still have to have a method of representing the entire quantity of atoms. 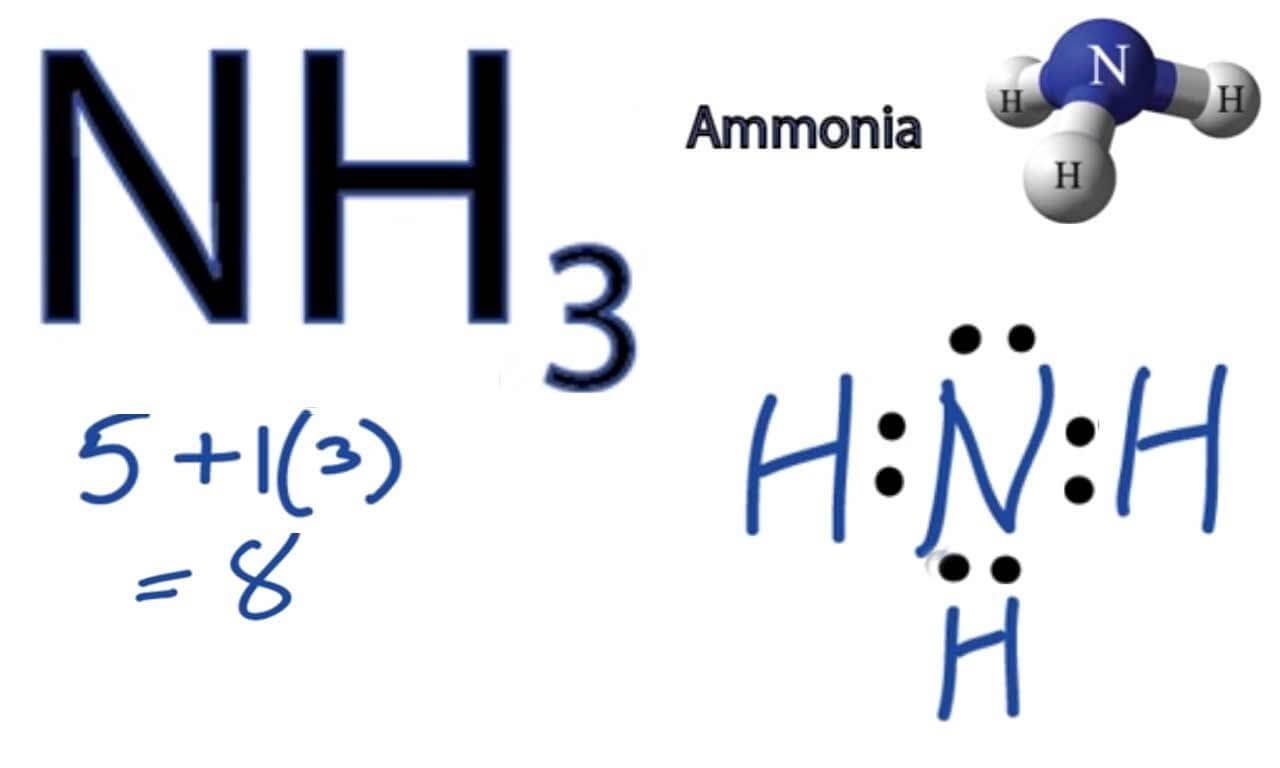
So why is the mole important in chemistry? It is important to have a unit of measurement that represents a large number of elementary entities, because when chemical changes occur they involve billions of atoms being rearranged. This means that there are 6.022×10 to the 23rd elementary entities found in one mole. This number is referred to as Avogadro’s number, which is 6.022×10^23. The number of elementary entities in one mole is equivalent to the number of atoms in 12 g of carbon 12. A mole is a unit of measurement for chemical substances, and it allows chemists or other scientists to determine how many elementary entities (molecules or atoms) are within a certain amount of a chemical substance. In order to understand why the molar mass of a substance is important, we will first need to define a mole. The molar mass of a chemical substance is the mass which is possessed by a single mole of that substance. The chemical properties of ammonia include ammonia having high stability, being combustible in air, and forming nitric oxide when combined with a platinum-rhodium catalyst at approximately 800☌. 
The molar mass of NH3 (ammonia) is 17.031 g per mole. Ammoniais a chemical compound that has the formula NH3, being made out of one nitrogen atom and three hydrogen atoms. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
